Chlorine has isotopes [math]Cl^{35}[/math] and [math]Cl^{37}[/math]. There are three [math]Cl^{35}[/math] isotopes for every one Cl isotope in a sample of chloride. What is the atomic mass of chlorine? - Quora

Calculate the percentage of the naturally occuring cl-35 and cl-37 that accounts for the atomic mass - Brainly.in

Atomic masses L.O.: Define the terms relative isotopic mass and relative atomic mass, based on the 12C scale; Calculate the relative atomic mass of. - ppt download
Naturally occuring chlorine is 75% Cl35 which and 25% Cl37. Calculate the average atomic mass of chlorine :– - Sarthaks eConnect | Largest Online Education Community

Calculate the atomic mass (average) of chlorine using the following data: `{:(,,% natural abunda... - YouTube









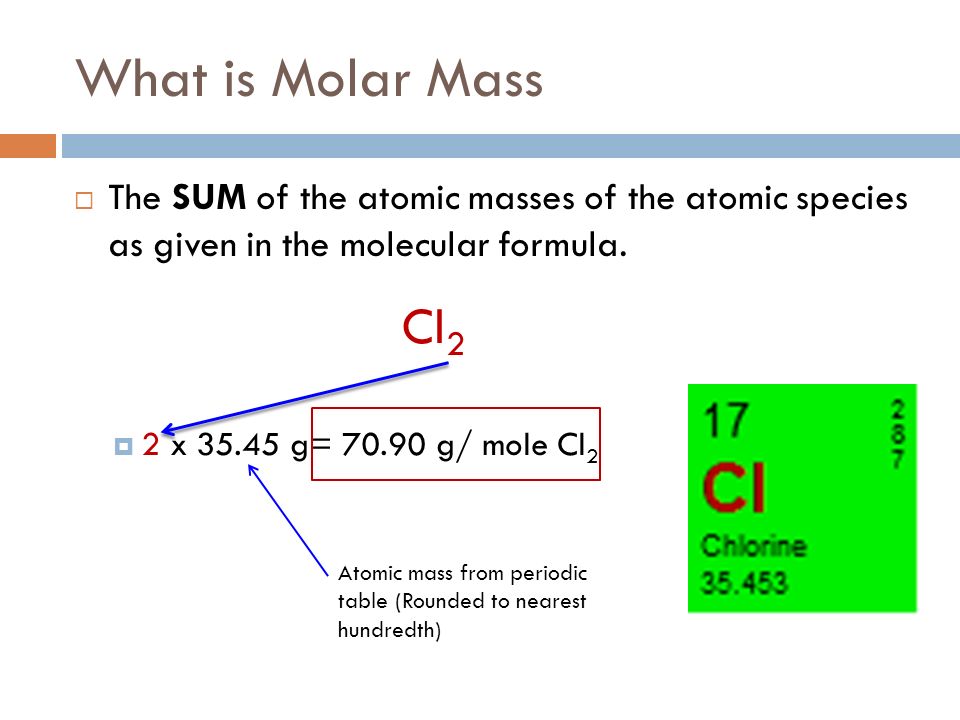
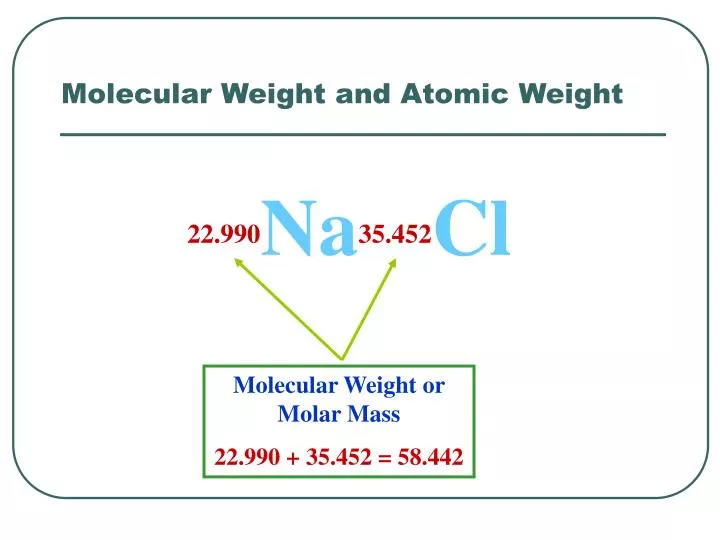

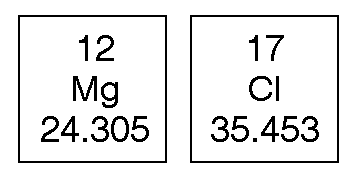
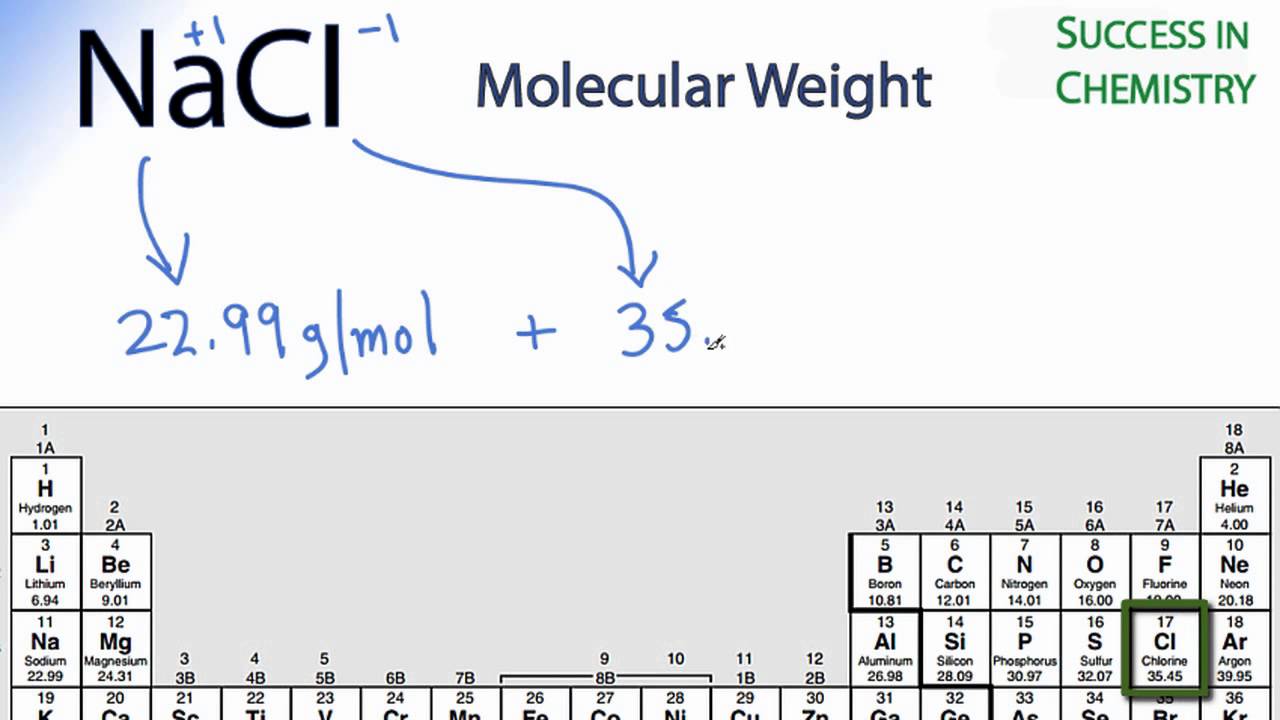
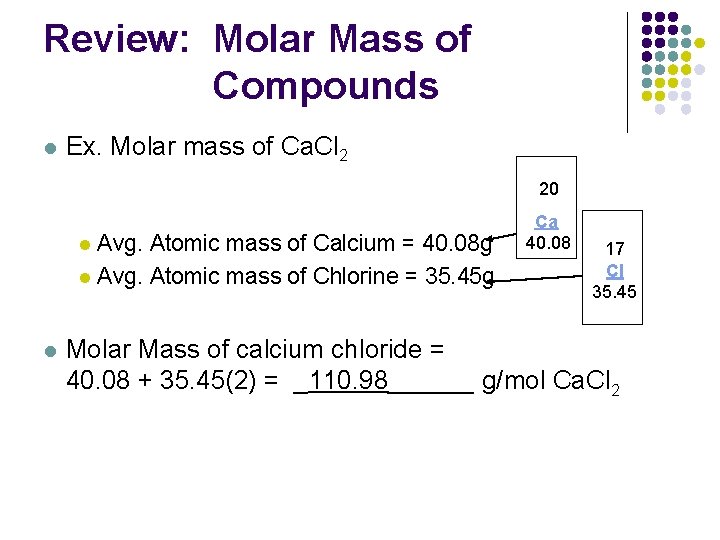




.PNG)



